Salinity is the amount of salt in water. It’s measured in parts per thousand, so if you have 100 ppt, that means there are 1 gram of salt dissolved into 1000 grams of water. If it’s too high or low you can do one thing to help deal with it: add more sodium chloride and/or remove some salts through filtering systems like reverse osmosis.
Salinity is one of the most important factors that affect the growth and survival of aquatic organisms. The effect of temperature on salinity is a complicated process, but it’s not impossible to deal with it.
As an aquarist, you’ve probably heard that temperature has an impact on salinity. However, since it is a difficult notion, I never really comprehended it.
So, how does salinity change as a result of temperature?
Specific gravity is used to determine the salinity of water. Temperature has a direct impact on this parameter. The density of saltwater rises or decreases as the temperature of the water changes. As a result, the specific gravity changes. However, there are ways to deal with this development.
Changes in salinity might be hazardous to your tank’s residents. It is preferable to address these modifications as soon as feasible. As a result, your tank will be a safe haven for many living species.
We’ve attempted to provide all you need to know about this subject. We discussed how to deal with a shift in salinity in your saltwater tank. Let’s get into the meat of the matter!
Does Temperature Affect Salinity? Mythbusting: Does Temperature Affect Salinity?
The metric for measuring the quantity of salt in water is salinity. It’s usually expressed in ppt (parts per thousand). It refers to the quantity of salt particles per 1000 water particles. Let’s say your tank’s salinity is 35 parts per thousand. It indicates that for every 1000 water particles, there are 35 salt particles.
The quantity of salt remains the same if no salt is added externally. So, what occurs in reality?
The shift is due to a change in specific gravity. As the temperature rises, the specific gravity drops. And as the temperature drops, so does the specific gravity. As a result, the salinity fluctuates.
When two samples with differing temperatures are compared, the warmer sample will have less salinity. The salinity of the cooler samples, on the other hand, will be higher.
Relationship between temperature and salinity
Salinity is determined by the specific gravity of saltwater, as we all know.
Relative density is another term for specific gravity. It is the ratio of a sample of saltwater at a certain temperature to the density of fresh water at the same temperature, according to the definition.
The water molecules in saltwater expand apart as the temperature increases. As a consequence, it takes up more room and is less dense. Meanwhile, salt stays unchanged. As a result, the salinity and specific gravity of the water drop.
When the temperature drops, however, the water molecules shrink. The density of water continues to rise, but the salt level remains constant. The specific gravity and salinity rise as a consequence.
Let’s look at an example of how specific gravity drops as temperature rises.
I maintained the salinity at 30.6ppt to see what would happen. I took measurements with this refractometer, which you can get on Amazon, after raising the temperature at regular intervals. The outcome was just as I had predicted.
| Temperature in degrees Celsius/degrees Fahrenheit | Specific Gravity at 30.6ppt Salinity |
| 7 degrees Celsius (44.6 degrees Fahrenheit) | 1.025 |
| 55.4°F (13°C) | 1.024 |
| 64.4°F (18°C) | 1.023 |
| 71.6°F (22°C) | 1.022 |
| 77°F (25°C) | 1.021 |
| The temperature is 28 degrees Celsius (82.4 degrees Fahrenheit). | 1.020 |
| 31 degrees Celsius (87.8 degrees Fahrenheit) | 1.019 |
| The temperature is 34 degrees Celsius (93.2 degrees Fahrenheit). | 1.018 |
| 37 degrees Celsius (98.6 degrees Fahrenheit) | 1.017 |
With rising temperature, you may witness a progressive drop in specific gravity. All of these figures seem to be a little perplexing, don’t they? We’ve also included a diagram to help you understand it better.
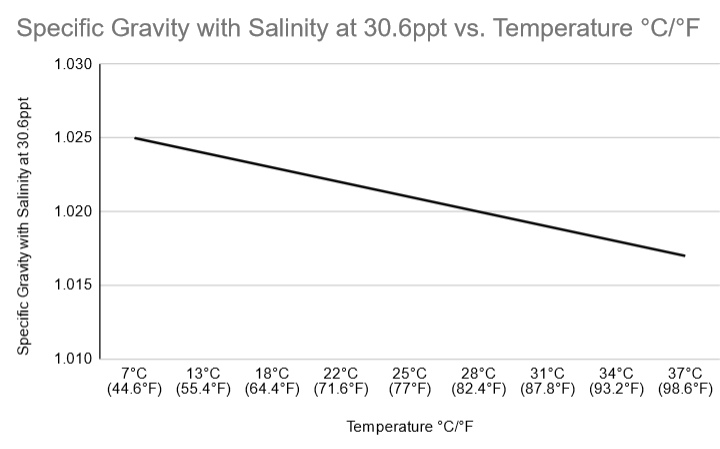
As you can see, as the temperature rises, the line descends. When a result, as the temperature rises, specific gravity decreases.
Evaporation’s Effect
However, there is a catch! Salinity levels are directly influenced by temperature. Evaporation occurs when water molecules begin to convert into vapor at a higher temperature.
The rate of evaporation increases as the temperature rises. Because when the molecules are heated, they travel quicker. Evaporation, on the other hand, may occur at lower temperatures.
That’s why you should use thermometers to check the temperature in your tank on a regular basis. Plants like frogbits may perish if the temperature rises too high.
For your aquarium, have a look at these digital thermometers. I used them in both my saltwater and freshwater aquariums.
Evaporation may, however, occur at lower temperatures. The water within the aquarium progressively evaporates, resulting in water vapor.

TigTag Aksorn is the source of this information.
The quantity of salt stays constant when the amount of water decreases. As a result, the quantity of salt in the water is increased. As a result, the salinity level rises.
But how can all of these adjustments be implemented without causing damage to the tank’s population?
How to Deal with the Salinity Shift
It’s hardly rocket science to deal with salinity shifts. It’s simple to do and just takes a little patience. It may also be used to adjust salinity when the temperature changes. However, here’s the rub: it’s not the most efficient method.
The salinity of the tank might rise or fall. However, you could try changing the water to maintain the salinity at its best. Keep in mind that nothing can be changed suddenly.
The fish will be startled by the sudden shift in water. Allow yourself plenty of time to replace the water in your tank. If you don’t, you’ll suffer the same fate as me. I was impatient when I first began this activity, and all of my fish died.
Because I needed to modify the salinity immediately, my whole tank’s population was in peril. So, let’s look at how you can effectively modify the salinity of your tank.
The salinity of my tank has risen.
In comparison to your aquarium, the notion of salinity in saltwater is completely different. Evaporation plays a significant part in this process. Here’s the deal: the salinity of water increases when the temperature rises due to evaporation. Furthermore, a rise in temperature may be the cause of your java fern’s death.
A drop in salinity is also caused by a drop in temperature. That is why we, as hobbyists, presume that ocean water has an average salinity level. It has a specific gravity of 1.026 and a 35ppt concentration.
But what can you do if the salinity in your tank surpasses this limit?
Is it true that changing the temperature lowers salinity?
You might want to increase the temperature to change the specific gravity. However, it contradicts the Evaporation’s Effect on salinity.
Because evaporating seawater raises the salinity of your prized aquarium. Furthermore, doing so will need a tremendous quantity of heat. The frog in your aquarium may perish as a result of the extreme heat.
Allowing the temperature to increase to that level might be detrimental to your plants, fish, and corals. It’s also one of the reasons why corals become dormant.
As a result, increasing the salinity by changing the temperature is impracticable. Maintaining an average temperature, on the other hand, is essential for maintaining optimal salinity.
Depending on your tank, the average temperature fluctuates. The temperature in your saltwater tank will not be the same as in your freshwater tank.
As you can see, it varies depending on the kind of tank. And here is where the aqueon vs. marineland dispute begins.
You may, however, utilize these dependable and easy-to-control aquarium heaters to regulate the temperature.
The size of your aquarium heater should be determined by the size of your tank.
Is it true that adding more water lowers salinity?
Yes, adding extra freshwater will dramatically lower the salinity level. Basically, you have two options for lowering the salinity of your water tank.
Method 1: Manually substituting freshwater for saltwater
To begin, take 10% of the tank water from your tank. After that, add the equal quantity of freshwater. When filling the tank, be sure to use reverse osmosis deionized water.
This RODI water is the cleanest kind of water available, with 0 percent ions. It’s the purest kind of water. Also, don’t forget to alter the temperature of the water you’re going to use. The water should be the same as in the tank.
Check the salinity of your tank water using your refractometer or hydrometer now. Repeat this method until it achieves its maximum potential.
Method 2: Automated Top-Off System
Repeating the water-replacement operation by hand might be tedious. It’s also time-consuming and monotonous. Most of us, let’s face it, don’t want to go to all of that trouble.
The good news is that there is a machine that can assist you in this procedure automatically. It’s known as the Auto Top Off System, or ATO for short.
It’s a mechanism that slowly fills your tank with fresh water. It intrinsically analyses your tank’s data and supplies the freshwater your tank requires. All you have to do is keep the RODI water in the ATO reservoir full.
You’ll never have to worry about your tank’s salinity increasing again, believe me. I’ve used several excellent ATO and have some recommendations for you.
You may acquire any of these and see how well your tank does.
The salinity of my tank has dropped.
The biggest challenge for many new saltwater enthusiasts is maintaining constant settings. I wasn’t an outlier, either. I’ve made a lot of errors in order to figure out what works best for my tank.
Even if you have all of the necessary tools, all you need is effort. The more work you put into tank maintenance, the better. However, no matter how hard you try, unexpected things might happen.
As an example, your tank’s salinity is being reduced. More salt is required to raise the salinity level. So, how do you know how much salt you’ll need?
To begin, measure the salinity (or specific gravity) of your seawater. It will assist you in determining the quantity of salt required to restore equilibrium. You may use our automated salt addition calculator to figure out how much salt you’ll need.
Another option is to do it by hand. Don’t worry about the arithmetic; I’ll assist you.
Let’s assume the salinity in a 300-gallon tank is 30ppt. This equates to 30 grams of salt per liter of water. You must now obtain 35 points each minute.
You’ll need 5g salt to raise 1L of 30ppt water to 35ppt.
Because 1 gallon equals 3.8L, it will need (3.8*5) =19g per gallon. Again, you’ll need (300*19) = 5700 grams = 5.7 kg of salt for a 300 gallon tank.
This isn’t entirely right, but it gives you a good idea of how much salt you’ll need.
Isn’t it therefore the most obvious thing to do to add the salt? But first, let me clarify something.
Is it Possible to Put Salt in a Tank Directly?
Salt should never be added directly to your aquarium. This will result in a massive water imbalance in the tank. It will cause your tank’s normal operation to be disrupted. It will also endanger the lives of the tank’s residents.
It’s acceptable if you’re establishing a fresh tank without any live organisms. That tank may be sprayed with salt. Never, ever, ever add salt to a crowded tank. It will be suicidal in nature.
So, what’s the solution to this issue of how to add salt to an aquarium?
Remove 10% of the aquarium’s water first. Then, into a huge bucket, add the equal quantity of RODI water. Then, in the RODI water, dissolve the appropriate quantity of salt. After that, set it to the same temperature as the aquarium water. Finally, fill the tank 1/2 gallon at a time with the salt-water combination. Continue doing so until the required salinity level is reached.
Different kinds of salt are required for various tank types. On the market, there are salts that have been specially prepared. You can find them here and purchase one for your prized tank.
You are the only one who understands your tank inside and out. As a result, choose your salt wisely. Because it has the potential to make or damage your priceless aquarium setup.
Are you certain the solution was successful?
In this post, we’ve looked at a variety of options. You now know how to decrease as well as raise the salinity of your tank. But how can you be so certain that everything went off without a hitch?
This is when salinity and specific gravity come into play. Hobbyists used to use a hydrometer to check these parameters back in the day. They are, however, quite unreliable.
The expense of a refractometer is the rationale for employing a hydrometer. The good news is that it is now within reach. When compared to a hydrometer, the precision of a refractometer is far higher. Here are several dependable refractometers that I believed performed well.
So, what’s the best way to utilize a refractometer?
Checking Salinity with a Refractometer [Step-by-Step Guide]
It’s not difficult to use a refractometer. Making a pond in a terrarium is nothing like this. You must first calibrate the refractometer before determining the salinity of your tank. To assist you, below is a step-by-step instruction.
Dripping RO Water on the Main Prism (Step 1 of 6)
Place a few drops of RODI water on top of the main prism first. The daylight plate may be opened to do this. Distilled water may be used instead of RODI water.
Close the plate and let the water to saturate the main prism’s whole surface. You must ensure that the surface is free of dry areas and bubbles.
Holding the Refractometer Towards the Light Source (Step 2 of 6)
Keep the main prism in place for 45 seconds once you’ve finished soaking it. After then, hold the instrument up to the light source. Then, using the eyepiece, you’ll see a circular field. Try changing the focus if the image isn’t clear.
The circular field should be blue in hue. The blue circular field indicates that the salinity is zero. If you don’t see anything other than blue, the refractometer has to be calibrated.
Calibrating using a Calibration Screw (Step 3 of 6)
You must gaze across the eyepiece to calibrate the refractometer. The calibration screw must then be adjusted. Adjust it until the blue and white fields are perfectly aligned at 0.
Soaking the Main Prism in Salt Water (Step 4 of 6)
It’s time to test the salinity now that your refractometer has been correctly calibrated. To begin, use a pipette on apply some tank water to the prism. Now, give the sample around 45 seconds to acclimatize to the ambient temperature.
Observing Through the Eyepiece (Step 5 of 6)
This stage is quite similar to the second. To examine the readings, you must look through the eyepiece. Take note of the blue portion’s bottom border and the white portion’s top limit. The difference between these two parts shows your saltwater’s salinity level.
Cleaning the Main Prism (Step 6 of 6)
Finally, distilled water should be used to clean the primary prism. You may also clean the prism with RODI water. After that, wipe the water away using a soft towel. You must ensure that it is completely clear of any water particles.
It’s critical to keep the salinity of your tank stable. It has the potential to make or ruin the whole ecology inside the tank. It’s always preferable to avoid a disaster before it occurs.
As a result, here are some suggestions for preventing salinity changes.
Tip 1: Only use fresh water to finish.
One thing we overlook is that it is the water that evaporates, not the salt. As the water volume decreases due to evaporation, the salinity rises. You must replenish the aquarium water with new water to reduce the salinity.
The salinity will grow if you add saltwater to compensate for evaporation. Because you have added additional salt to the saltwater that already exists. Using fresh water to fill the tank, on the other hand, will maintain the salinity constant.
Replacing the evaporated water with an equivalent amount of purified fresh water, on the other hand, will maintain the salinity where you want it.
Another thing to keep in mind is the amount of time between top-offs. To prevent salinity changes, it should be completed quickly. Consider this procedure as one of your everyday responsibilities.
Tip 2: Matching the Added Salt Water’s Salinity
What measures can you take now that you’ve added salt water? To begin, compare the salinity of the water to be added to the salinity of the tank water.
A word of advice: don’t trust the salinity level shortly after you’ve finished adding new saltwater. Allow the solution to settle for at least one night. Check it once you’ve cooked the freshly mixed saltwater.
It’s possible that the salinity measurement will change somewhat from what it was before. Because the salt will have had a time to mingle and stabilize by then. If there are any variations, either salt or freshwater should be added as needed.
It’s now time to cautiously pour it into the tank.
Tip 3: Checking and adjusting the temperature on a regular basis
It may seem simple to check your tank’s temperature for any abnormalities on a regular basis. However, it takes time and effort to master.
Checking the temperature of the tank on a regular basis is essential to avoid any unforseen occurrences. If there are any deviations from the predicted value, make the necessary adjustments to your aquarium heater.
FAQs
When it comes to fish, what is the ideal salinity range?
The majority of fish want to be in a salinity range that is optimal for them. It ranges from 1.023 to 1.028. This does not imply that the fish within this range are safe. For fish, the optimal salinity level is 1.026 0.001.
What happens if the salinity is too high for corals?
It is beneficial for coral development if the concentration of dissolved salt is greater. Corals will be harmed by salinity levels over 1.027, but they will not perish. Anything beyond 1.030, on the other hand, is very harmful, and you should endeavor to lower it as soon as possible.
Is 1.030 Sp. Gravity excessive?
Salinity levels as high as 1.030 Sp.gravity are unsuitable for a saltwater aquarium. It’s recommended to keep the salinity at approximately 1.026. Otherwise, corals and fish will be harmed.
Bottomline
Your saltwater tank’s salinity is a critical parameter. It is inextricably linked to changes in temperature. There are a lot of misunderstandings about this connection. However, we attempted to resolve this ambiguity once and for all.
That concludes our contribution. We’ve done our best to explain how salinity is affected by temperature.
If you have any more questions, please leave a comment below. Best of luck!
The “how does density affect salinity” is a question that has been asked many times. The answer to this question is that it depends on the type of salt, and how much water there is in the solution.
Related Tags
- how does evaporation affect salinity
- how does temperature affect saturation
- how does temperature affect the density of seawater
- how does depth affect salinity
- how does temperature and salinity affect the density of water

